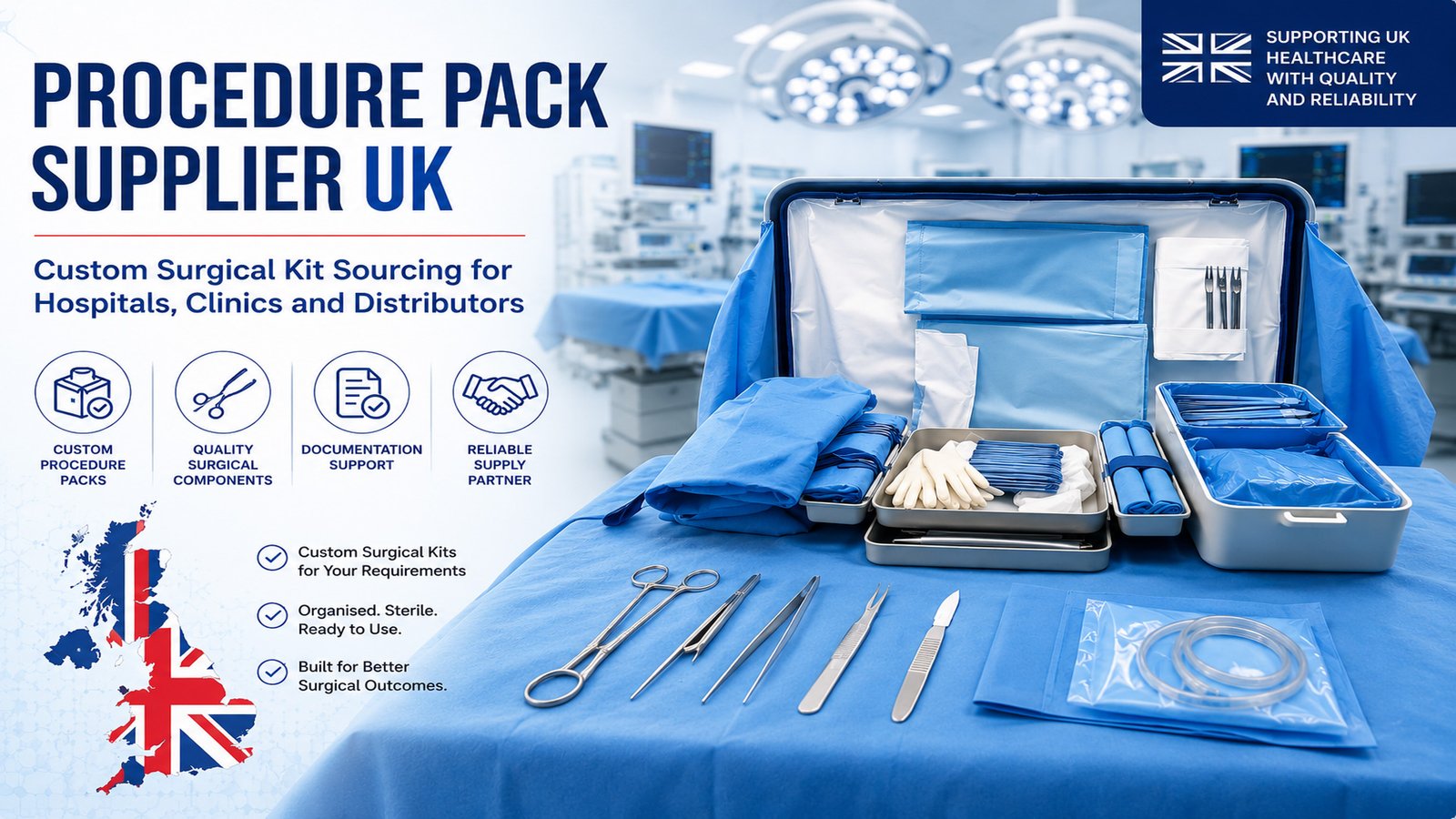
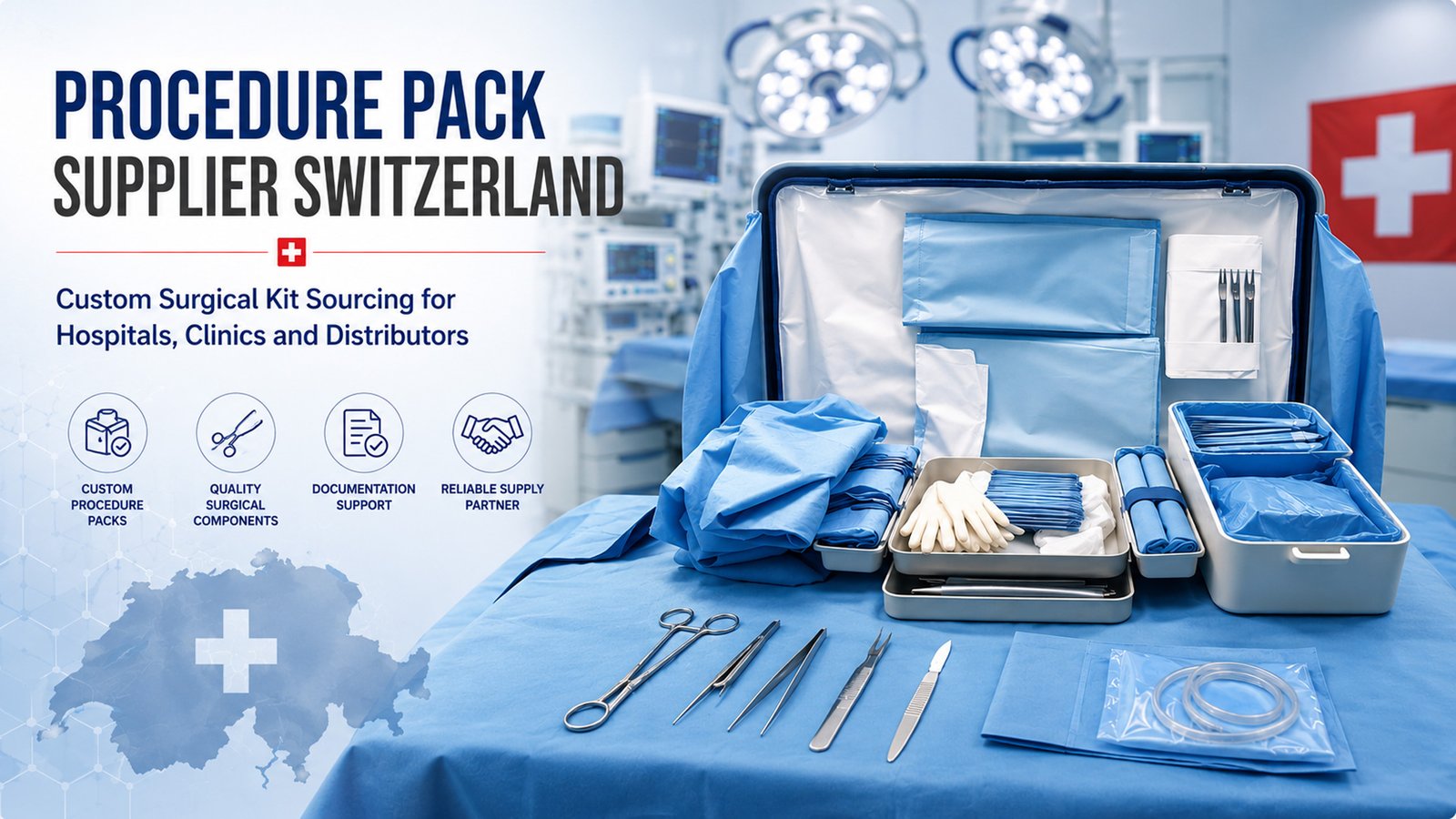
At Cosil Instruments Company, we are committed to delivering reliable surgical, dental, beauty, and electrosurgical instruments and accessories with consistent performance, safety, and customer satisfaction. Our quality approach is built on controlled processes, trained personnel, and continual improvement.
Our Commitments
Customer & Regulatory Focus
We aim to meet applicable customer, statutory, and regulatory requirements for our products and markets.
Quality Management System
We maintain a structured Quality Management System aligned with medical device industry expectations and risk-based thinking. For relevant electrosurgical devices, referenced standards include ISO 13485 and risk management practices (e.g., ISO 14971) as applicable.
Process Control & Traceability
We emphasize:
Incoming inspection of raw materials/components
In-process and final inspection criteria
Lot/batch traceability and documentation appropriate to the product type
Product Safety & Risk Management
We identify and control product risks through documented procedures, corrective actions, and preventive actions (CAPA), and continuous monitoring of feedback/complaints.
Supplier Quality
We evaluate suppliers to ensure materials and components meet defined specifications and quality requirements.
Training & Competence
We train our team to follow standardized work instructions, quality checks, and safety practices.
Continuous Improvement
We use customer feedback, audits, and performance indicators to improve product quality, on-time delivery, and service responsiveness.
Quality Objectives
Maintain consistent product conformity to agreed specifications
Reduce nonconformities through preventive controls
Improve customer satisfaction through responsive support and reliable dispatch
Contact (Quality/Compliance): sales@cosilinstruments.com